Collection method: In-Person Blood Draw
Measures
IgG
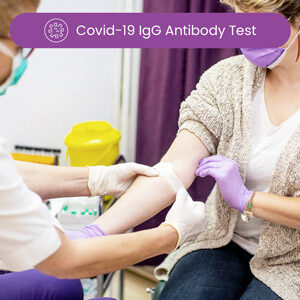
Covid-19 IgG Antibody Test
$85.00
Select your testing service:
FSA/HSA accounts accepted
Manufactured by Ortho Clinical Diagnostics, this was the first quantitative COVID-19 IgG antibody test to receive FDA Authorization. This test targets the S1 spike protein of SARS-CoV-2 IgG antibodies and was calibrated to the World Health Organization’s International Standards. With 100% specificity, this test is designed to give clinicians and public health leaders a standard tool to measure antibody response to SARS-CoV-2. The MyLabsDirect COVID-19 IgG quantitative antibody report presents a reactive (positive) or non-reactive (negative) analysis for the presence of antibodies found in a specific patient sample. The report also presents the manufacturer’s reference range data for the assay. The reference range for this assay is 17.8 to 200 Units per mL. A patient sample with a reported value that falls between 17.8 and 200 or is greater than 200 U/mL is determined reactive (positive) for the detection of the S1 spike protein of SARS-CoV-2 IgG antibodies. A patient sample with a reported value below 17.8 is determined non-reactive (negative) for the detection of the S1 spike protein of SARS-CoV-2 IgG antibodies.
How It Works
1.
2.
3.